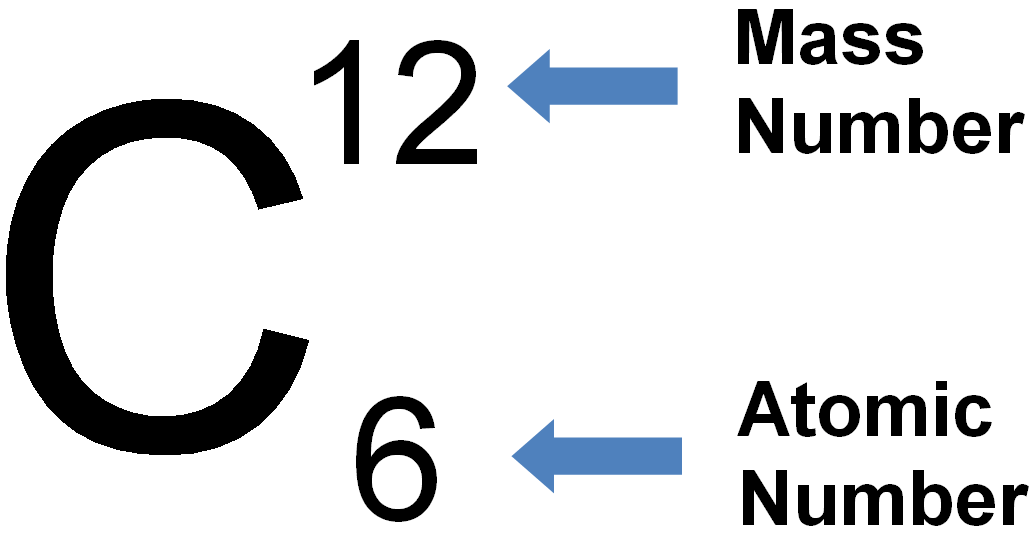 
The number of protons determines an element’s atomic number, which scientists use to distinguish one element from another. The reason they do not is that the electrons that surround all atoms are negatively charged and negative charges repel each other.Ītoms of each element contain a characteristic number of protons and electrons. With all this empty space, one might ask why so-called solid objects do not just pass through one another. In these atoms, the positive and negative charges cancel each other out, leading to an atom with no net charge.Īccounting for the sizes of protons, neutrons, and electrons, most of the atom's volume-greater than 99 percent-is empty space. In uncharged, neutral atoms, the number of electrons orbiting the nucleus is equal to the number of protons inside the nucleus. Although not significant contributors to mass, electrons do contribute greatly to the atom’s charge, as each electron has a negative charge equal to the proton's positive charge. Therefore, when considering atomic mass, it is customary to ignore the mass of any electrons and calculate the atom’s mass based on the number of protons and neutrons alone. Hence, they do not contribute much to an element’s overall atomic mass. Electrons are much smaller in mass than protons, weighing only 9.11 × 10 -28 grams, or about 1/1800 of an atomic mass unit. Therefore, the number of neutrons in an atom contributes significantly to its mass, but not to its charge. A proton is positively charged whereas, a neutron is uncharged. Although similar in mass, protons and neutrons differ in their electric charge. Scientists arbitrarily define this amount of mass as one atomic mass unit (amu) or one Dalton, as Table 2.2 shows. Protons and neutrons have approximately the same mass, about 1.67 × 10 -24 grams. Atoms are made up of protons and neutrons located within the nucleus, with electrons in orbitals surrounding the nucleus. The most common isotope of hydrogen (H) is the only exception and is made of one proton and one electron with no neutrons.įigure 2.2 Elements, such as helium, depicted here, are made up of atoms. Atoms contain protons, electrons, and neutrons, among other subatomic particles. The atom's outermost region holds its electrons in orbit around the nucleus, as Figure 2.2 illustrates. We cannot break down gold atoms into anything smaller while still retaining the properties of gold.Īn atom is composed of two regions: the nucleus, which is in the atom's center and contains protons and neutrons. A gold coin is simply a very large number of gold atoms molded into the shape of a coin and contains small amounts of other elements known as impurities. For example, one gold atom has all of the properties of gold, like its chemical reactivity. An atom is the smallest unit of matter that retains all of the element's chemical properties. To understand how elements come together, we must first discuss the element's smallest component or building block, the atom. In spite of their differences in abundance, all elements and the chemical reactions between them obey the same chemical and physical laws regardless of whether they are a part of the living or nonliving world. For example, the atmosphere is rich in nitrogen and oxygen but contains little carbon and hydrogen, while the earth’s crust, although it contains oxygen and a small amount of hydrogen, has little nitrogen and carbon. In the nonliving world, elements are found in different proportions, and some elements common to living organisms are relatively rare on the earth as a whole, as Table 2.1 shows. The four elements common to all living organisms are oxygen (O), carbon (C), hydrogen (H), and nitrogen (N). For example, the symbol for sodium is Na, referring to natrium, the Latin word for sodium. Other elements’ chemical symbols derive from their Latin names. Some elements follow the English term for the element, such as C for carbon and Ca for calcium. The remaining elements are unstable and require scientists to synthesize them in laboratories.Įach element is designated by its chemical symbol, which is a single capital letter or, when the first letter is already “taken” by another element, a combination of two letters. There are 118 elements, but only 98 occur naturally. Elements are unique forms of matter with specific chemical and physical properties that cannot break down into smaller substances by ordinary chemical reactions. Matter is any substance that occupies space and has mass. Explain the ways in which naturally occurring elements combine to create molecules, cells, tissues, organ systems, and organismsĪt its most fundamental level, life is made up of matter.Compare the ways in which electrons can be donated or shared between atoms.Describe the interrelationship between protons, neutrons, and electrons. 
By the end of this section, you will be able to do the following:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
